Displaying items by tag: pmc
How Your Center Can Help Prevent Ebola's Spread

As the Ebola outbreak has spread from West Africa to isolated cases in parts of Europe and now the United States, there has been no shortage of panic and paranoia among Westerners—even those working in the health care fields.
Still, it is an established and repeated fact that Ebola's spread can be prevented simply by following proper safety procedures in a medical setting. An informational page on Ebola published at the World Health Organization's (WHO) website points to the sad reality of Ebola's largely preventable spread:
Health-care workers have frequently been infected while treating patients with suspected or confirmed EVD [Ebola Virus Disease]. This has occurred through close contact with patients when infection control precautions are not strictly practiced.1
As those involved in the life-saving work of pregnancy help in the medical setting, how can you protect yourself, your clinic, and your clients from this deadly virus?
The answer starts with educating yourself, your staff and volunteers, and your clients as to how Ebola spreads, which, again according to WHO, involves the following:
Ebola... spreads through human-to-human transmission via direct contact (through broken skin or mucous membranes) with the blood, secretions, organs or other bodily fluids of infected people, and with surfaces and materials (e.g. bedding, clothing) contaminated with these fluids.2
Consider the functions of your Pregnancy Help Medical Center or Clinic.
- Could you come into contact with any bodily fluid when handling a urine pregnancy test?
- What about collecting a specimen for STD/STI testing?
- Broken skin that can be exposed and contacted while conducting an ultrasound?
With these points of contact posing risks in light of the Ebola virus—in addition to several diseases that are much more likely to spread—make sure your center is paying special attention to policies such as Universal Precautions as defined by the Centers for Disease Control (CDC) and OSHA Regulations.
Under Universal Precautions, blood and certain body fluids of all patients are considered potentially infectious for HIV, (Hepatitis B), and other blood-borne pathogens. Universal Precautions involve the routine wearing of gloves, other protective clothing, hand washing, and such infection control measures that are designed to place a barrier between potentially infectious blood or body fluids and employees.3
For more information on how you can keep your center protected from the spread of viral infection, check out Heartbeat International's Medical Essentials for Pregnancy Help Organizations©.
1. "Ebola virus disease," World Health Organization, http://www.who.int/mediacentre/factsheets/fs103/en/ (accessed Oct. 17, 2014).
2. Ibid.
3. Medical Essentials for Pregnancy Help Organizations©, Heartbeat International, 2014, Part IV, page 4.
The Buzz from the Medical Tracks
We love hearing from you in the pregnancy help medical community.
Here's a sampling of what attendees said about the 2014 Heartbeat International Conference in Charleston, South Carolina. The Conference featured 78 workshops, including six each in the Medical Matters and Ultrasound Advancement tracks, in addition to two medically focused in-depth all-day workshop sessions and a special keynote from John Bruchalski, M.D., founder of Tepeyac Family Center & Divine Mercy Center.
1. "I feel much more confident after these workshops and webinars. Thank you!"
Jo-Ellen O'Keefe, Pregnancy Help and Information Center (Ectopic Pregnancy, Audrey Stout RDMS)
2. "Very interesting and groundbreaking information – this will change things!"
Reagan Nielson, Vitae Foundation (Update on Abortion Pill Reversal, Dr. Delgado)
3. "Dr. Delgado's presentation was so very exciting! It was interest peaking, informative and empowering. I'm grateful for his work and research regarding the APR. (Abortion Pill Reversal)"
Jamie Koser, Heartline Pregnancy Center (Update on Abortion Pill Reversal, Dr. Delgado)
4. "Very creative teaching! Lots of light bulbs went off!"
Cheryl Didrekson, Kimberly Home, Inc. (Flipping Uterus, Roxanne Ertel RDMS)
5. Amazing presentation! I have been scanning for 12 years and finally understood the anatomy of the uterus on U/S! Thank you!"
Jennifer Snowden, New Beginnings Women's Center (Flipping Uterus, Roxanne Ertel RDMS)
6. "Super excellent."
Katherine Niemiec, Family Life Services Clinic and Pregnancy Center (Flipping Uterus, Roxanne Ertel RDMS)
7. "Thank you! This was the most enlightening session of the week and worth the entire trip to learn these nuggets."
Patti White, New Beginnings (Flipping Uterus, Roxanne Ertel RDMS)
When am I Due? Why Should I be Concerned?
Healthy Pregnancy/Healthy Baby Series: Part 1
By Helen Risse RN MSN
 If you work with pregnant women, you have a great opportunity to improve birth outcomes. When a new client visits your pregnancy help organization, this may be the only contact you have with her.
If you work with pregnant women, you have a great opportunity to improve birth outcomes. When a new client visits your pregnancy help organization, this may be the only contact you have with her.
What should she be sure to know before she leaves you? Does she know her due date? Will you be telling her based on the first day of her last menstrual period?
It is important to define due date and term pregnancy. Remind your new mother that her due date is really a due time that looks at two weeks before to two weeks after that date as being "term". Many people still think of pregnancy in terms of nine months. Explain that pregnancy is defined as 40 weeks or 10 lunar months.
At the end of 2013, the American College of Obstetrics and Gynecology (ACOG) redefined the meaning of "term".
- Early term: Between 37 weeks, 0 days and 38 weeks, 6 days.
- Full term: Between 39 weeks, 0 days and 40 weeks, 6 days.
- Later term: Between 41 weeks, 0 days and 41 weeks, 6 days.
- Post term: Between 42 weeks, 0 days and beyond.
Research has noted that the brain of a baby at 35 weeks, 0 days grows in size by two-thirds in the following four weeks.
Research has noted that although the weight of a baby may look normal, babies born before 39 weeks are sleepier babies. These babies do not latch and suckle as well as babies born at 39 weeks 0 days. They have more problems with higher bilirubin levels. These concerns can lead to serious consequences. The choice of an elective delivery date must factor in these findings.
Women should also be taught the signs of preterm labor. Teach women about contractions. Explain what they may feel and describe those symptoms that should put them on alert.
Describe contractions as feeling like:
- Menstrual cramps
- Low, dull backache
- Pelvic pressure—feeling that the baby is pushing down
- Heavy feeling in your pelvis (pelvic congestion)
- Stomach cramps—with or without diarrhea
- The abdomen may tighten or get firm then relax or soften
Describe vaginal discharge or bleeding:
- An increase or change in your normal vaginal discharge
- Red, brown or pink discharge or spotting
Describe water breaks:
- Gush of fluid from vagina
- Slow trickle of fluid from vagina-(panties feel wet and you don't know why)
General feeling that something is not right.
What should she do if she thinks she may be having preterm labor? Below are some guidelines you may discuss with your Medical Director to develop a policy/procedure for your center.
- Empty bladder
- Drink 1-2 glasses of water or juice (no caffeine, sugar, or sports drinks)
- Lie down on your side and time the contractions from the beginning of one to the beginning of the next. Write down when the contractions start.
If the contractions are coming more than every 15 minutes or 4-6 in an hour, call your doctor.
It is important to stay well hydrated. Dehydration can often cause a woman to experience contractions.
Women should know the risks that increase concerns for preterm labor. Women who are at greatest risk for preterm labor are those who have had a previous preterm birth, as well as those who are pregnant with multiples, and those with certain abnormalities of their uterus or cervix.
Other risk factors include smoking, drinking alcohol, using illegal drugs, domestic violence, including physical, sexual or emotional abuse, or lack of support.
Additional risks factors related to her health include infections, including urinary tract infections, sexually transmitted infections, diabetes, high blood pressure, second trimester bleeding from the vagina, being underweight before pregnancy, obesity, and a short time period between pregnancies.
When asked if there is anything that can be done to prevent preterm labor, tell a woman to:
- See her doctor early and regularly during pregnancy,
- Eat nutritious meals and snacks,
- Drink at least 8 glasses of water, juice or milk every day, and
- Avoid cigarettes, alcohol, drugs and medications not prescribed by her doctor.
If you have one visit from a pregnant woman and pass on this information, you may contribute to an improved outcome. Every extra day her baby is in a healthy intrauterine environment is positive for the development of her baby, which in turn can be a big help to a new mother.
References
Spong CY. Defining "Term" Pregnancy: Recommendations From the Defining "Term" Pregnancy Workgroup. JAMA. 2013;309(23):2445-2446. doi:10.1001/jama.2013.62
ACOG Clinical Guidelines: Definition of term pregnancy. Committee Opinion No. 579. American College of Obstetricians and Gynecologists. Obstet Gynecol 2013;122:1139–40.35
Go the Full 40 Campaign tool Kit :http://www.health4mom.org/pregnancy/healthy_pregnancyo
The last weeks of pregnancy count: July 5th, 2012l Kit: http://newsmomsneed.marchofdimes.com/?tag=brain-development
At the Heart of What it Takes to go Mobile
by Michele Chadwick, Director of Operations, ICU Mobile
 Heartbeat's Betty McDowell gave an encouraging message in her presentation at the 2014 Heartbeat International Conference, which came down to a single phrase: I see you. The God of the universe acknowledges and recognizes you. You are valid.
Heartbeat's Betty McDowell gave an encouraging message in her presentation at the 2014 Heartbeat International Conference, which came down to a single phrase: I see you. The God of the universe acknowledges and recognizes you. You are valid.
At ICU Mobile, our name and mobile ultrasound ministry (ICU, think "I See You") was born out of a call to proactively reach out to help mothers see their babies in the womb, identifying that every life is valid—sometimes for the first time in a mother's heart.
Revealing and affirming life is at the heart of all our ministries, serving a vital, essential role in life-affirming work. When a pregnancy center provides medical ultrasound imaging, its representatives affirm that the act of visibly revealing life is essential to the ability to fully inform a pregnant mother of the life she carries.
Going mobile medically brings this vital life-imaging service to mothers outside of the center. At ICU Mobile, we believe that at the heart of an accomplished mobile medical clinic, as in the pregnancy center, is a commitment to offer services with faith, wisdom, and experience as our guide. We are committed to standing strongly on the strategic operating principle of extending the reach of our services for women into the –community—in the community of others.
A mobile medical clinic, when operating well, is a tool with the capability of joining a community together under the umbrella of life, drawing pregnancy resource centers together with the church and other community and organizational resources, in support of each other in the service of women.
Key factors that help encourage this united front include neutral branding, a process to provide a full continuum of care for the client, operating from each other's strengths through shared service models, and encouraging good stewardship through shared resources.
 Pregnancy help expert Kirk Walden, in his book, "The Wall" (and who also presented at the Heartbeat Conference!) makes the case for uniting the community to serve women. He asks the question, "What happens if we're the first choice?" The "we" in this question is key. The "we" is all of us working together to be a mother's first choice. A mobile medical clinic can be the "vehicle" (no pun intended) for building this unity.
Pregnancy help expert Kirk Walden, in his book, "The Wall" (and who also presented at the Heartbeat Conference!) makes the case for uniting the community to serve women. He asks the question, "What happens if we're the first choice?" The "we" in this question is key. The "we" is all of us working together to be a mother's first choice. A mobile medical clinic can be the "vehicle" (no pun intended) for building this unity.
Mobile operations in joining others together can become one of the most effective ways to reach abortion-minded women. Mobile services provide an effective way to expand your reach without having to build a satellite office, expand internally, or require more space, and it allows a center the flexibility to move to locations to adjust to demographic changes and population shifts.
Further, going mobile avoids the issue of no-show appointments at your center, as well as helping to balance the number of clients seeking social support services and those seeking medical services. Going mobile alleviates the expense of renovation or expansion of services in your center, while providing opportunities to build network supports with other pregnancy help organizations in the community surrounding the center.
Each of these considerations factor into the thought process of going mobile in a community.
When considering the possibility of adding a mobile medical clinic, it is first important—as with adding any medical service—to recognize the seriousness of the endeavor and requires the highest level of commitment to a professional quality medical experience for the client, as it is visible and the first contact with a mother.
Equally important for an organization to note is that going mobile is a unique approach that influences how a center operates, affecting the procedural processes that are needed to accommodate a moving center. A board, leaders, staff, and the connected community must pray and seek wisdom and recognition of the call to serve in this mission capacity.
We recommend you seek others who already operate a mobile medical clinic to learn about effective methods of operation, strategies that work, and the significance of working together in community to serve. It is God who builds these mobile communities, so it will be God who lays the possibility and provides the opportunity for success.
As Kirk Walden asked at the Heartbeat Conference, "What can we do to join hands?" Mobile done in community with others will join hands, and as we are blessed to say at ICU Mobile, it joins hands for little feet.
Here are some practical questions and steps toward adding mobile services:
- Do you have a need to reach women in multiple locations who would benefit from the ability to change locations daily?
- Is your board and staff ready to take a logistically challenging step that would involve a comprehensive team effort?
- Contact ICU mobile and ask questions that would be important for you to consider when incorporating a new program.
- Once the board has the buy-in, meet with top donors to survey their interest in the project.
- Investigate friends and "influencers" on local college campuses to assess their interest.
Healthcare Worker
Serving as a nurse or other medical professional in a pregnancy help medical clinic (PHMC) can be of great benefit to clients and your organization. Physicians can serve as medical directors, which allows organizations to offer medical support such as ultrasound, STI/STD testing, prenatal care, and more. Nurses and sonographers, with the appropriate training, can provide ultrasound scans, pregnancy tests, and more. Even outside individual medical clinics and healthcare providers can join the Abortion Pill Rescue Network to help women who are hoping to save their children after starting a chemical abortion as prescribing providers or hotline consultants.
Heartbeat International has a fantastic array of courses available online through the Heartbeat Academy just for nurses in need of CEUs as well. Here is a simple way to gain additional skills and knowledge valuable to your position and ministry in the pregnancy help medical clinic while obtaining the Contact Hours you need to keep your license active.
Opportunities for training or supporting the pregnancy help movement for healthcare workers:
- Join the Abortion Pill Rescue Network
- Medical Matters (monthly eNewsletter free to affiliates)
- Archives (affiliates only)
- Medical Essentials Manual
- Online CEU Opportunities
- LOVE Approach Ultrasound Clinical (LAUC) Training
- Online Medical Training
- Abortion Pill Reversal Training
Welcome to Heartbeat!
  |
Heartbeat International has the most expansive network
of pregnancy help in the world!



Heartbeat's Life-Saving Vision is a world where every new life is welcomed and children are nurtured within strong families, according to God’s Plan, so that abortion is unthinkable.
Heartbeat's Life-Saving Mission is to Reach and Rescue as many lives as possible through an effective global network of life-affirming pregnancy help that Renews communities for LIFE.
To achieve our mission, we:
Advancing Life-Affirming Pregnancy Help Worldwide
To become an affiliate of Heartbeat International, you must review and agree to abide by the Heartbeat Principles and the Commitment of Care and Competence:
 |
 |

- Heartbeat International does not promote abortion, abortifacients, or contraceptives.
- Heartbeat International does not promote birth control (devices or medications) for family planning, population control, or health issues, including disease prevention.
- Heartbeat International does promote God's Plan for our sexuality: marriage between one man and one woman, sexual intimacy, children, unconditional/unselfish love, and relationship with God must go together.
- Heartbeat International does promote sexual integrity/sexual purity before marriage and sexual integrity faithfulness within marriage.
- All Heartbeat International policies and materials are consistent with Biblical principles and with orthodox Christian (Catholic, Protestant, and Orthodox) ethical principles and teaching on the dignity of the human person and sanctity of human life.
Not all pregnancy centers offer medical services. If you have questions about the services offered at this pregnancy center, please ask to speak to a center representative.
Welcome to Heartbeat!
Experts to Help! Pro-Life Maternal-Fetal Medicine Docs
Two important resources for your center and medical director
 |
by Susan Dammann RN, Medical Specialist
Dedicated to the care and preservation of both mother and fetus in every pregnancy, Pro-Life Maternal Fetal Medicine (MFM) is an association of pro-life doctors of like mind to AAPLOG (American Association of Pro-Life Obstetricians and Gynecologists).
This unique organization represents a contingent of life-minded Maternal Fetal Medicine (MFM) practitioners. (MFM is a subspecialty of Obstetrics and Gynecology dealing with all matters that can affect the health of a mother or fetus from before conception to the postpartum period.)
Members of Pro-Life MFM are also affiliated with the Society for Maternal-Fetal Medicine, a special interest group of the American College of Obstetrics and Gynecology who have received additional training and performed research in the care and management of pregnant women and fetuses.
The Role of Pro-Life MFM Practitioners
MFM professionals are specialists in high-risk pregnancy situations, and hold a uniquely expert place in relation to rank-and-file OB/GYN physicians or oncologists. MFM specialists are involved in guiding the management of medical and surgical complications a mother may encounter during pregnancy.
MFM specialists also provide diagnosis and management of medical and surgical conditions for the fetus. Care may include in utero treatment, modification of delivery timing or mode, and facilitation and coordination of care for the infant after delivery.
When a client presents in your center with a negative maternal or fetal diagnosis, there are alternatives to help ensure the survival of both mother and baby. For example, many women with a breast cancer diagnosis have carried their pregnancies to term and done better than women who abort.
Are you looking for a pro-life Maternal-Fetal Medicine specialist in your area? Visit Pro-Life MFM’s physician directory.
Hear from a Pro-Life MFM Expert
Dr. Murphy Goodwin, a well-known pro-life maternal fetal medicine specialist, wrote an excellent article called Medicalizing Abortion Decisions. Dr. Goodwin, whose obstetric practice in the Los Angeles area has been the largest in the United States for most of the last 15 years, serves many of the high-risk deliveries in the area.
While describing five cases of successful delivery where a mother had abortion recommended to her, Dr. Goodwin states that because of the dangerous combination of an ambivalent attitude toward the developing human in the medical community and fear of liability issues (owing to the unbalanced legal burden of informed consent and “wrongful birth”), physicians are often not providing readily available information that could affect their patients’ judgment regarding abortion when that mother has a major medical problem in pregnancy or any medical problem.
To suggest or recommend that abortion is the safest route carries no such responsibility, as there is no set legal precedent for a physician’s liability in a case where abortion was recommended on supposed medical grounds—even if that recommendation was subsequently found to be baseless or misrepresented.
Tragically, as Dr. Goodwin points out, “There is no counterweight to ‘wrongful birth.’ There is no ‘wrongful abortion.’
These are two helpful resources you’ll want to keep handy and make available to your medical director!
An Informed Look: A New Tool for Sharing the Truth
by Laura Strietmann, Associate Director, Pregnancy Center East, Cincinnati, Ohio
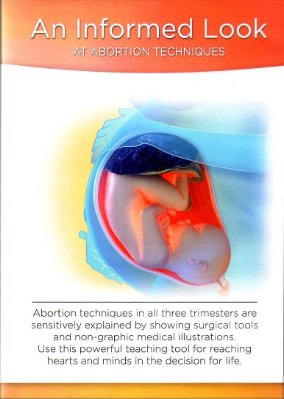
When I began serving clients in crisis at Pregnancy Center East in Cincinnati, Ohio over 7 years ago, I noticed that besides time, love, and an ultrasound, there was another powerful tool in assisting a woman in the choice of life for her unborn baby.
This was an outdated grainy VHS tape, entitled Abortion Techniques. Non-graphic in content, but real and compassionate, each time this tape was viewed by a client considering abortion, she left with a different mindset. The client usually moved from being abortion-vulnerable to choosing life for her baby.
Carol Everett, a former abortion clinic owner and operator, turned pro-life warrior, had filmed Abortion Techniques in 1993. It was a 25-minute video showing abortion through illustrations, actual tools, and Carol’s personal testimony. Many centers throughout the country use this video in teaching pregnant women the realities of abortion. Today, abortion has been made to seem as if it is equivalent to having a mole removed, as a “necessary” aspect of healthcare. Abortion Techniques showed how far these perceptions are from reality. One day several years ago, while working at PCE, our copy of the video broke. After searching for an updated replacement for this worn tape, I discovered that the industry lacked a current video with the most recent abortion methods sensitively presented in such an effective manner.
I decided to contact Carol Everett, now very busy as an internationally known pro-life author, speaker, and lobbyist for the state of Texas. Had she thought of making a more updated version of the video? Did she realize the number of babies’ lives saved by this tape? Would she see it was time to make a new film? Several months of persistent emails, Facebook inbox messages, and finally a phone conversation before Carol conditionally agreed to the project.
Having never met in person, only through internet and phone lines, Carol promised that if I could secure funding for the project, she would journey to PCE and make a new video. She would generously assign PCE the rights to the video as a means for fundraising. Through the generosity of the Ruth J. and Robert A. Conway Foundation, PCE was able to secure the funds to film the new version. This was not the answer I thought I would receive in my initial inquiries, but it was an incredible opportunity to affect the lives of the babies at PCE and now throughout the country.
Carol traveled to Cincinnati and Greg Schlueter, a Catholic moviemaker, and staff member for the Diocese of Toledo, OH filmed the video. On the afternoon of February 15, 2013 just as filming was wrapping up at PCE our doorbell rang. With a CLOSED sign on the door, two women still rang our bell desperate for help. As I opened the door and they noticed all of the filming equipment, the client begged for assistance. Stepping inside the Center, the client shared she was already well into her second trimester, but had finally just told her sister, who then found the Center and brought her for help. The situation was sensitive and the expecting mom was also post abortive.
As the young pregnant client and her sister described the crisis, Carol rounded the corner and heard the story. Right there in the lobby of PCE Carol listened to this young client and through a beautiful conversation offered so much understanding and hope. Carol counseled the client with sincerity and love. This client stayed in PCE’s care throughout her pregnancy and a perfect baby was delivered this summer. Mom is overjoyed with how everything has worked out. This was the first miracle of life from the updated video being brought to reality.
An Informed Look at Abortion Techniques is the new 14-minute modern life-saving tool. Using modern colors and graphics Mertz Design Studios completed this version to also include information on Plan B as well as RU-486 abortions. The initial launch of the video took place in April 2013 at the Heartbeat International Conference, selling over 100 copies to centers as far away as Alaska, Africa, Austria, and Germany.
After viewing the video in Dallas at the Conference, Janet Morana, Executive Director of Priests for Life, shared, “Every pregnancy center should be showing this movie to clients.” Reviews from center directors throughout the country are calling it “powerful,” the “best tool next to an ultrasound in reaching hearts and minds in the decision for life.”
On the day I write this article one absolutely abortion determined mom sat in PCE with a volunteer and viewed An Informed Look. The decision for life was made then and there while watching the video, even before the ultrasound. The client was astounded by the reality of abortion procedures. What happens to her body as well as the unborn baby’s was shocking to say the least. Even in a non-graphic manner, seeing how an abortion is performed is devastating enough. When making the biggest decision of her life a woman deserves to have complete information. Carol’s testimony is riveting.
Hearing the sorrow of a post-abortive women provides another enormous window to the reality of abortion. Viewing this film allows a woman, in the words of Carol “To make her decision fully informed.”
We hope to get this tool in the hands of thousands, including pregnancy resource centers, high school educators, and politicians. We will be distributing the video at future Catholic and pro-life conferences. Please visit us in Washington, D.C. during The March for Life convention. The video will continue to be sold through www.HeritageHouse.com, as well as directly through the Center, at 513-321-3100.
If you would like to connect with Laura about presenting the video at any future appropriate meetings or conferences please contact her This email address is being protected from spambots. You need JavaScript enabled to view it. or 513-321-3100.
Abortion-Breast Cancer Link Explodes in Asia
 |
by Joel Brind, Ph.D.
A new systematic review and meta-analysis of abortion and breast cancer (ABC link) in China, was just published in November, 2013 in the prestigious, peer-reviewed international cancer journal, “Cancer Causes and Control”. It showed that the overall risk of developing breast cancer among women who had one or more induced abortions was significantly increased by 44%. In this meta-analysis (a study of studies in which results from many studies are pooled), Dr. Yubei Huang et al. combined all 36 studies that have been published through 2012 on the ABC link in China.
Also in peer-reviewed journals in 2013, Dr. Ramchandra Kamath et al. reported an odds ratio (a measure of relative risk) of 6.38 and Dr. A.S. Bhadoria et al. reported a relative risk of 5.03, i.e., a 5-fold—or 403%--increased risk of getting breast cancer among Indian women who have had any abortions. Not only are these relative risks much stronger than had been reported anywhere before (e.g., the 1.44 reported by Huang et al. in China and the 1.3 reported by my colleagues and I in our worldwide meta-analysis of 1996), but also in 2013, Dr. S. Jabeen and colleagues reported a relative risk of 20.62 among women in Bangladesh!
These new Asian studies change the game in ABC link research, and should completely abolish any credibility of the "politically correct" dictum of the US National Cancer Institute (NCI; a federal agency like the IRS and the NSA) that the ABC link is nonexistent.
Several reasons for this can be enumerated:
- The Huang meta-analysis reproduces and validates our findings from 1996, even showing a slightly stronger link (1.44 v. 1.3, respectively), and on entirely different populations of women (Chinese women since 1988 v. worldwide women from 1957-1996, respectively).
- The Huang meta-analysis also demonstrated what is called a "dose effect", i.e., two abortions increase the risk more than one abortion (76% risk increase with two or more abortions), and three abortions increase the risk even more (89% risk increase with three or more abortions). Risk factors that show such a clear dose effect have more credibility.
- The new Asian studies—especially those from the subcontinent—put the final nail in the coffin of the main argument used to discredit the ABC link, variously called the "response bias" or "recall bias" or "reporting bias" argument. The argument goes like this. Due to social stigma that is attached to having an induced abortion, healthy women are more likely to deny prior abortions in their medical history study questionnaire than are women who've developed breast cancer. Hence, the argument goes, it would erroneously appear that abortion is more frequent among women who have had breast cancer.
Although no credible evidence for this response bias hypothesis has ever been presented (and plenty of good evidence against it) in ABC link research, the NCI and others have continually cited it as if it were a matter of fact, to deny the reality of the ABC link. The fact is that such response bias is only even plausible when the relative risk is relatively low, such as around 1.5. But such bias becomes extremely implausible when the relative risk is strong—e.g. 5 or 6 or more. Thus, while one might attempt to explain how some women with breast cancer might be more or less inclined to report their history of induced abortion, the numbers from India and Bangladesh are just too overwhelming: In the Jabeen study, 262 breast cancer patients were compared to 262 healthy (control) women. It was found that 231 of the patients (88%) had had any abortions, whereas only 70 out of the 262 healthy women (27%) had had any abortions!
It is important to note that the Jabeen data does raise the question as to why there should be such a strong link in Bangladesh, if abortion should have the same effect on women everywhere The answer is straightforward: In Bangladesh, breast cancer is still rare because a)early marriage and childbearing—the best known protection against breast cancer—is nearly universal; b) breastfeeding (also a protective factor against breast cancer) is also nearly universal, as is c) the lack of alcohol consumption (a known risk factor). Consequently, there's not much in Bangladesh besides abortion to cause breast cancer, so it really stands out.
It is really frightening when you start doing the math on the impact of abortion on a population of over a billion women—in India and China alone: Just a 2% lifetime risk of breast cancer due to abortion—a very conservative estimate—means upwards of 10 million women getting breast cancer, and millions dying from it. Welcome to the real war on women.
Joel Brind, Ph.D. is a Professor of Human Biology and Endocrinology at Baruch College, City University of New York; Co-founder of the Breast Cancer Prevention Institute, Somerville, NJ; and member of the Heartbeat International Medical Advisory Council.
Maintaining Competency through Continuing Education
By Connie Ambrecht RDMS, CMB
 |
It was very exciting when your Pregnancy Resource Center (PRC) changed to a Pregnancy Medical Clinic (PMC). I am certain you can recall the enthusiastic atmosphere that filled the center as you reached your goal of changing from having a focus on material assistance to focusing on being able to answer the question; “Am I pregnant?” through medical imaging. Providing confirmation of pregnancy was finally a goal actualized!
Looking back at the steps that were taken, everyone on the team participated in training that was a necessary component to adding medical services. From the Board of Directors, the Executive Director and the Clinic or Nurse Manager, every aspect of the PRC was educated about the change. Our team at Sonography Now participated in the change at over 500 PRC to PMC transitions and our responsibility to you remains. Our role as an education company is to keep ourselves educated and pass that along to you. Our team attends approximately 6-8 conferences per year to keep abreast of trends that impact your clinical setting.
For many of you, that initial investment in getting to know what was needed for a PRC to become a PMC was the start and the finish to your education as it relates to being a clinic. Let’s review what is necessary for the medical personnel in particular to remain strong for the PMC. Then let’s consider: What does each member of the team need to do to remain strong for the PMC we serve?
For those such as NARAL Pro-Choice America who would like to see our doors close, they state things like: “CPCs are a growing threat to women's health. They often lie to women about abortion and birth control. They'll do anything to scare a woman away from choosing legal abortion.” They consider this a challenge to address.
We don’t believe there is a need to feel a panic in addressing such accusations, instead we feel it is wiser to be methodical about how you keep your entire organization educated. We want you to be empowered to discuss how and why you stay strong. In considering a methodical approach to your team’s education, there are a few things to ask… Who needs to be educated? How would we get such education? Where do we get the funding? Where can we go? Do we have to go anywhere? Who decides who needs what? There are so many legitimate things to consider.
This article is to assist you in building a plan for continuing education as it relates to your team, as well as providing talking points to aid in community relations.
As we begin, I’d like to outline a few facts: you provide medical services, sonography and maybe SDI testing, so that’s what makes you a clinic…embrace it. As a clinic, the public has certain expectations…be aware of those expectations. With those expectations comes the responsibility to provide your medical services at a level that meets standards as established by professional organizations.
Do you have an awareness of who those organizations are? Association of Women’s Health, Obstetric and Neonatal Nurses (AWHONN), American Nurses Association (ANA), and the Society of Diagnostic Medical Sonographers (SDMS)? Each of these organizations has guidelines and/or recommendations for continuing education. Let’s look at each one, create a talking point and build a plan to implement.
• According to AWHONN, the RN who performs sonography “should demonstrate educational and clinical expertise in obstetrics….”
• In the new Code of Ethics for Nurses with Interpretive Statements, the ANA clearly states in Provision 5 that “the nurse owes the same duties to self as to others, including the responsibility to preserve integrity and safety, to maintain competence, and to continue personal and professional growth.” Provision 7 states that “the nurse participates in the advancement of the profession through contributions to practice, education, administration, and knowledge development.”
• The SDMS clearly requires all sonographers, registered or unregistered, under Section II, to “maintain continued competence through lifelong learning, which includes continuing education, acquisition of specialty specific credentials and re-credentialing.”
If you take the three professional organizations above and build a talking point you might be ready to state; “As a clinic we find it is a priority to maintain competency, build skills and knowledge, because we value keeping up with medical standards.”
Your approach to building an education plan will vary from clinic to clinic. Annual Conferences such as the Heartbeat International Conference can build your medical team as well as others who attend. As education organizations, Sonography Now and Equip Leaders Now are committed to maintaining a large library of courses for the Pregnancy Medical Clinic that can grow your entire team. We recognize the importance of keeping up on trends, building the team's knowledge base and providing quality care and focus on that aspect of your services.
To quote Maya Angelou, “If we know better we do better”. We know you need continuing education in order to do better. Our obligation to you is to stay informed and pass it along. We look forward to serving you this year in regards to your education needs.
Connie Ambrecht RDMS CMB and her husband Dan founded Sonography Now in 2002. Connie has been published in professional journals, has spoken at national conferences. Sonography Now has provided training to physicians, nurses, physicians assistants, nurse practitioners, midwives, radiologic technologists, as well as cross-training other sonography specialists.







